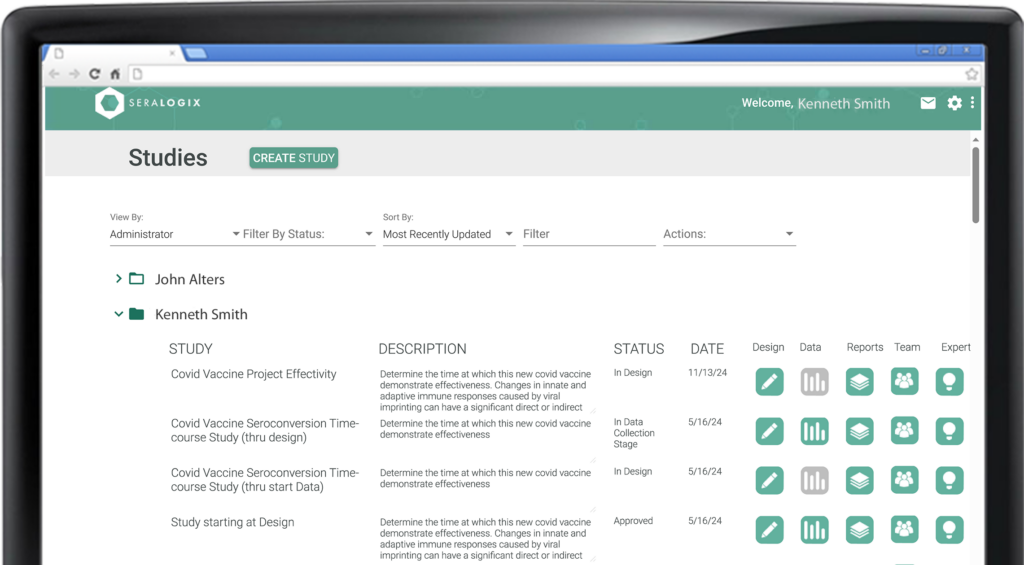
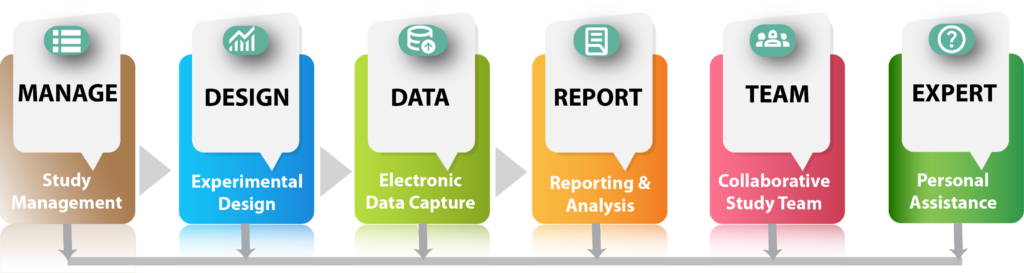
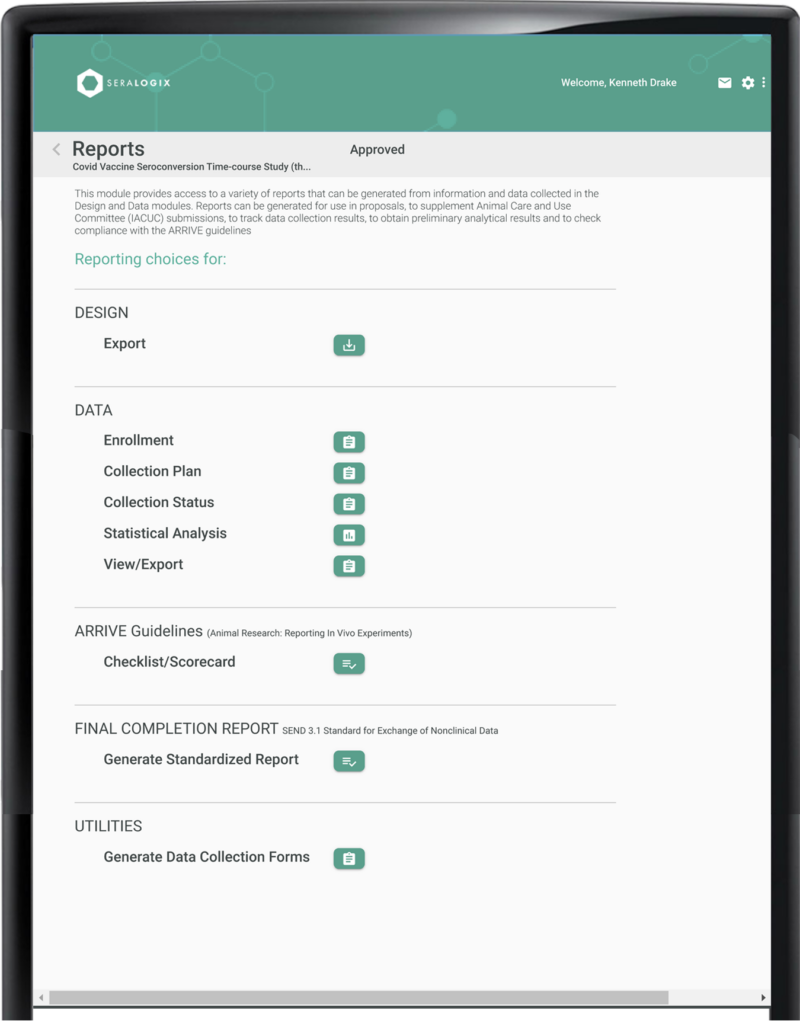
The Reports Module streamlines every aspect of your study’s reporting needs. From experimental design and subject enrollment to data collection plans, summary statistical analysis, and data export files, every essential document is automatically created at the touch of a button. The module also includes a thorough ARRIVE guidelines compliance checklist and a SEND 2.1 Standards-compliant final completion report (in development), ensuring your study meets the highest standards of accuracy, transparency and reproducibility.
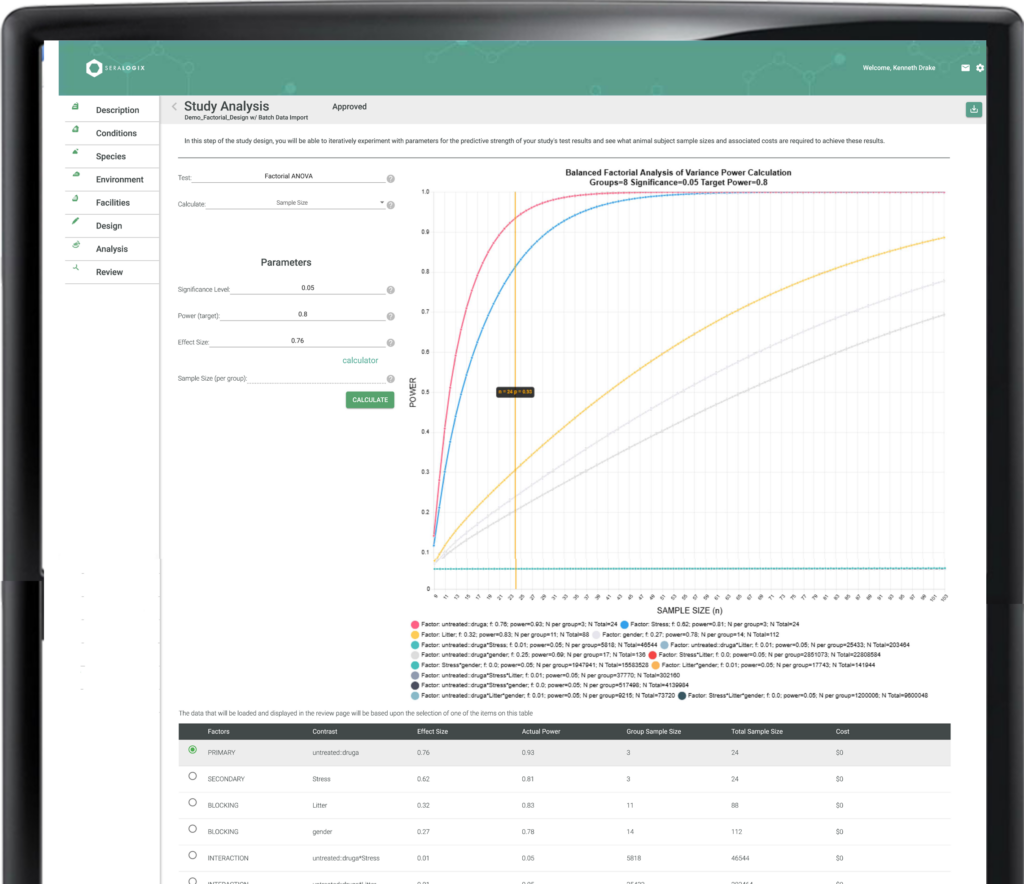
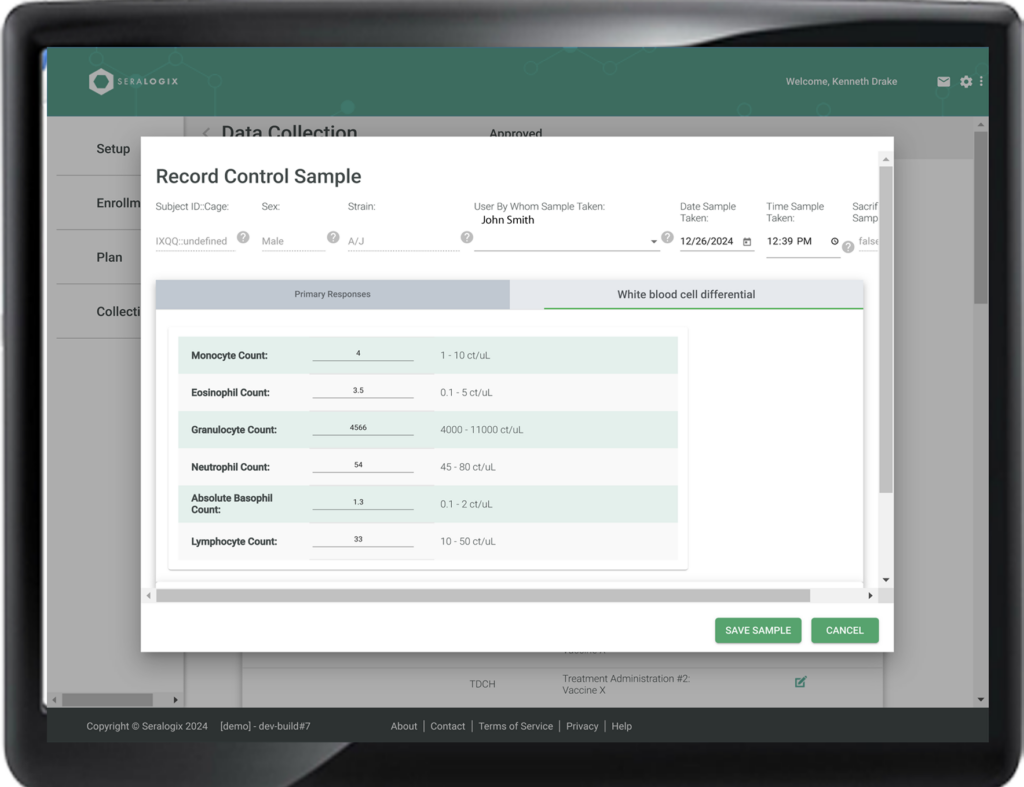
The Experimental Design Report details groups, conditions, treatments, environmental factors, outcome response metrics, timelines, and sample size derivations based on power and significance criteria—perfect for rapid IACUC submissions or proposal inclusions. The Collection Plan offers a precise, longitudinal schedule of data collection and treatment events, while the Collection Status Report provides real-time insights, highlighting data collection progress and notifying you of any late events. The Statistical Analysis Report delivers immediate summaries of collected data, helping you quickly identify outliers and confirm data quality. Additionally, the ARRIVE Guidelines Report ensures your design and results meet reporting standards, supporting compliance with ease.